Now, thanks to a new drug, we may not have that worry anymore.
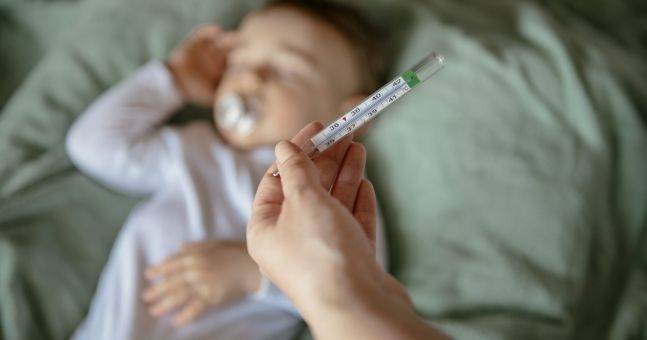
RSV, or respiratory syncytial virus, is a childhood illness which sees an infant with a fever, cough, runny nose and trouble breathing.
According to the HSE, almost all children get RSV by the time they are two and it typically spreads in winter and early spring.
Most symptoms of RSV are mild, such as a runny nose, coughing and sneezing and people usually recover within two to three weeks, but babies and toddlers are most vulnerable.
The Food and Drug Administration (FDA) in the US has now approved a new medication that can help prevent the virus in babies and toddlers.
The drug is called Beyfortus (nirsevimab) which is made by AstraZeneca and can be given to newborns and babies during or entering their first RSV season.
The medication can also be given to toddlers up to 24-months who are heading into their second RSV season.
"Many parents have had an experience with RSV in their lives that hasn't been great," Gigi Chawla, MD, MHA, chief of general paediatrics at Children's Minnesota, said.
"I think the utility of this type of treatment will be seen by parents differently than perhaps other vaccine offerings we've had.
"
The American Academy of Pediatrics (AAP) has also followed the FDA approval and has recommended that all infants should receive the treatment to protect them against severe RSV.
Along with the AAP and the FDA, the Centres for Disease Control and Prevention (CDC) have also recommended a single dose of the medication for all infants younger than 8 months born during or entering their first RSV season as well as babies and toddlers between 8 and 19-months who are at high risk, the same was the AAP recommendation.
"Paediatricians are sadly familiar with the dangers of RSV and the devastating consequences facing some families," Sandy Chung, MD, FAAP said in a news release.
"We are eager to offer all infants this protection and urge federal officials to see that it is made available and affordable in all communities."